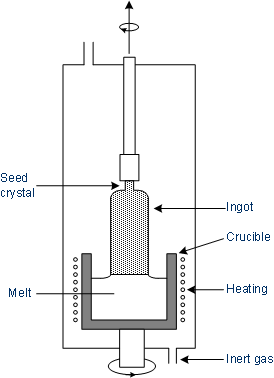
The polycrystalline silicon, as it is present after the zone cleaning, is melted in a quartz crucible nearly above the melting point of silicon. Now dopants (e.g. boron or phosphorus) can be added to the melt to achieve appropriate electrical characteristics of the single crystal.
A seed crystal (a perfect single crystal) on a rotating rod is brought to the surface of the silicon melt. This seed crystal pretends the orientation of the silicon crystal. In contact with the seed crystal, the melt overtakes its crystal structure. The fact that the crucible temperature is only slightly above the melting point of silicon, the melt solidifies immediately on the seed and the crystal grows.
The seed is slowly pulled upward with constant rotation, while there is constant contact with the melt. The crucible turns in the opposite direction of the seed crystal. A constant temperature of the melt is essential to ensure a steady growth. The diameter of the single crystal is determined by the drawing speed, which provides 2 to 25 cm/h. The higher the drawing speed, the thinner the crystal. The entire apparatus is located in a controlled atmosphere, so that no oxidation of silicon can take place.
The disadvantage of this procedure is that the melt is accumulated with dopants during the process, since the dopants are more solubly in the melt than in the solid state. Thus the dopant concentration along the silicon rod is not constant. Also impurities or metals can dissolve from the crucible and built into the crystal.
The advantages of this method are the lower costs, and the ability to produce larger wafer sizes as in flot-zone processes.
Illustration of the Czochralski process